SOLUBILITY
-Solubility: The maximum amount of solute that dissolves in a given amount of solvent at a constant temperature.
-Solubility is usually expressed in grams of solute per 100 grams of solvent at a specified temperature.
-Knowing the solubility of a substance can help you classify solutions based on how much solute they contain.
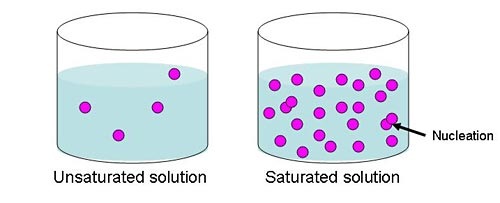
-Saturated: A solution that contains as much solute as the solvent can hold at a given temperature. When a solution is saturated, the solvent is "filled" with solute. If you add any more solute, it will not dissolve.
-Solubility: The maximum amount of solute that dissolves in a given amount of solvent at a constant temperature.
-Solubility is usually expressed in grams of solute per 100 grams of solvent at a specified temperature.
-Knowing the solubility of a substance can help you classify solutions based on how much solute they contain.
-Saturated: A solution that contains as much solute as the solvent can hold at a given temperature. When a solution is saturated, the solvent is "filled" with solute. If you add any more solute, it will not dissolve.
-Unsaturated: A solution that has less than the maximum amount of solute that can be dissolved. For example, if you sweeten your tea with sugar and the sugar dissolves you know it is an unsaturated solution. As long as the amount of solute is less than the solubility at that temperature, the solution is unsaturated.
-Supersaturated: A supersaturated solution is one that contains more solute than it can normally hold at a given temperature. These solutions are very unstable. If you put even a little bit more than the solution can hold, it may rapidly deposit out of solution.
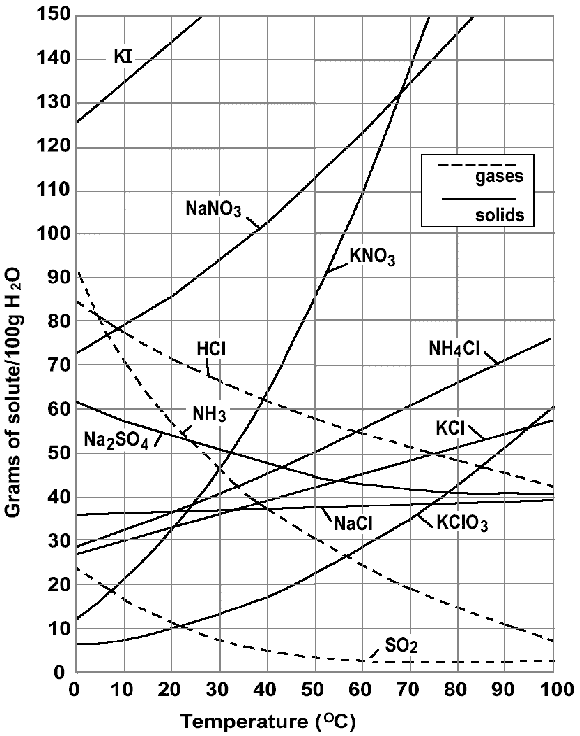
SOLUBILITY CURVE
-Supersaturated: A supersaturated solution is one that contains more solute than it can normally hold at a given temperature. These solutions are very unstable. If you put even a little bit more than the solution can hold, it may rapidly deposit out of solution.
SOLUBILITY CURVE
-These curves are very simple.
-Basically if you are looking at a line, say NaCl, if a point is on the line, it is saturated. Below the line is unsaturated, and above the line is supersaturated. Its that simple.
-You might be asked a question like, what type of saturation would the point (20, 30) have for HCl?
- Your answer would be unsaturated.
CONCENTRATION
-Concentration of a solution: The amount of solute dissolved in a specified amount of solution.
-Percent by Volume: percent by volume = Volume of solute / Volume of solution X 100%
-Percent by Mass: This is more useful when the solute is a solid.
Percent by mass = mass of solute / mass of solution X 100%
-Molarity: The number of moles of a solute dissolved per liter of solution. Moles are the amount of a substance that contains approximately 6.02 X 10 ^ 23.
Molarity = moles of solute / Liters of solution
QUESTIONS
1. On a solubility curve, how can you tell if a point is saturated? (see student solutions)
2. What is solubility usually expressed in? (see student solutions)
3. Find the saturation of (30,30) for NaCl.
4. What is concentration?
-Basically if you are looking at a line, say NaCl, if a point is on the line, it is saturated. Below the line is unsaturated, and above the line is supersaturated. Its that simple.
-You might be asked a question like, what type of saturation would the point (20, 30) have for HCl?
- Your answer would be unsaturated.
CONCENTRATION
-Concentration of a solution: The amount of solute dissolved in a specified amount of solution.
-Percent by Volume: percent by volume = Volume of solute / Volume of solution X 100%
-Percent by Mass: This is more useful when the solute is a solid.
Percent by mass = mass of solute / mass of solution X 100%
-Molarity: The number of moles of a solute dissolved per liter of solution. Moles are the amount of a substance that contains approximately 6.02 X 10 ^ 23.
Molarity = moles of solute / Liters of solution
QUESTIONS
1. On a solubility curve, how can you tell if a point is saturated? (see student solutions)
2. What is solubility usually expressed in? (see student solutions)
3. Find the saturation of (30,30) for NaCl.
4. What is concentration?
The light bulb application is solubility. You can show solubility with a glass of water. It is a neat little thing that we can see in life if we look hard enough.