CHEMICAL BONDS
-Chemical bonds are exactly what they say. A bond between things at the chemical level.
-There are two main types of chemical bonds. Ionic and Covalent.
-Ionic Bonds: Elements that do not have complete sets of valence electrons tend to react. When t they react they achieve electron configurations similar to noble gases. One way that elements achieve stable electron configurations is through the transfer of electrons between atoms. What basically happens when two elements are trying to reach stable configurations is they share electrons with each other until everything is equal.
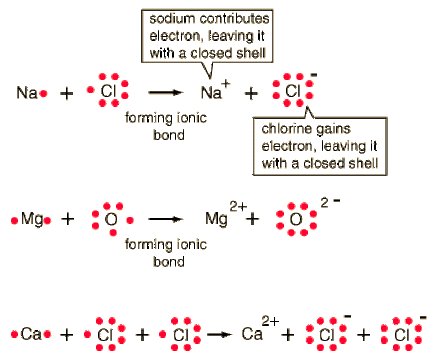
-When an atom gains or loses an electron, the number of protons is no longer equal to the number of electrons. The charge on the atom is not balanced and the atom is not neutral. An atom with a net positive or negative charge is called an ion. The charge on an ion is represented by plus or minus sign.
-Formation of ionic bonds: A particle with a negative charge will attract a particle with a positive charge. Just like magnets. When an anion (ion with a negative charge) and a cation (ion with a positive charge) are near each other a chemical bond forms between them. A chemical bond is the force that holds atoms or ions together as a unit. An Ionic Bond is the force that holds cations and anions together as a unit. An ionic bond forms when electrons are transferred from one atom to another.
-Chemical bonds are exactly what they say. A bond between things at the chemical level.
-There are two main types of chemical bonds. Ionic and Covalent.
-Ionic Bonds: Elements that do not have complete sets of valence electrons tend to react. When t they react they achieve electron configurations similar to noble gases. One way that elements achieve stable electron configurations is through the transfer of electrons between atoms. What basically happens when two elements are trying to reach stable configurations is they share electrons with each other until everything is equal.
-When an atom gains or loses an electron, the number of protons is no longer equal to the number of electrons. The charge on the atom is not balanced and the atom is not neutral. An atom with a net positive or negative charge is called an ion. The charge on an ion is represented by plus or minus sign.
-Formation of ionic bonds: A particle with a negative charge will attract a particle with a positive charge. Just like magnets. When an anion (ion with a negative charge) and a cation (ion with a positive charge) are near each other a chemical bond forms between them. A chemical bond is the force that holds atoms or ions together as a unit. An Ionic Bond is the force that holds cations and anions together as a unit. An ionic bond forms when electrons are transferred from one atom to another.
-Covalent Bonds: Atoms held together by sharing electrons are joined by a covalent bond.
-Covalent bonds are between nonmetals only.
-Molecules: particles formed by covalent bonds.
-The attractions between the shared electrons and protons in each nucleus hold the atoms together in a covalent bond.
-Multiple covalent bonds: When tow atoms have three pairs of electrons, it is a triple bond. When two atoms share two pairs of electrons, it is a double bond.
-Polar Covalent Bond: a covalent bond in which electrons are not shared equally. When atoms form a polar covalent bond, the atom with the greater attraction for electrons has a partial negative charge. the other atom has a partial positive charge.
-The type of atoms in a molecule and its shape are factors that determine weather a molecule is polar or non polar.
NAMING COMPOUNDS
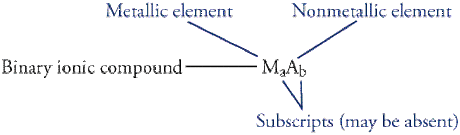
-Binary: A compound made from only two elements is a binary compound. Naming binary ionic compounds is easy. They have a predictable pattern: the name of the cation followed by the name of the anion.
-Polyatomic: A covalently bonded group of atoms that have a positive or negative charge and act as a unit. poly means many. most simple polyatomic ions are anions.
-Molecular: The name and formula of a molecular compound describe the type and number of atoms in a molecule of the compound. The most metallic element appears first in the name. The name of the second element is changed to end in the suffix -ide. for example: Carbon Dioxide.
WRITING CHEMICAL FORMULAS
-Binary and Polyatomic: Place the symbol of the cation first, followed by the symbol of the anion. Use subscripts to show the ratio of the ions in the compound. Because all compounds are neutral, all total charges on the cations and anions must add up to zero.
-Covalent bonds are between nonmetals only.
-Molecules: particles formed by covalent bonds.
-The attractions between the shared electrons and protons in each nucleus hold the atoms together in a covalent bond.
-Multiple covalent bonds: When tow atoms have three pairs of electrons, it is a triple bond. When two atoms share two pairs of electrons, it is a double bond.
-Polar Covalent Bond: a covalent bond in which electrons are not shared equally. When atoms form a polar covalent bond, the atom with the greater attraction for electrons has a partial negative charge. the other atom has a partial positive charge.
-The type of atoms in a molecule and its shape are factors that determine weather a molecule is polar or non polar.
NAMING COMPOUNDS
-Binary: A compound made from only two elements is a binary compound. Naming binary ionic compounds is easy. They have a predictable pattern: the name of the cation followed by the name of the anion.
-Polyatomic: A covalently bonded group of atoms that have a positive or negative charge and act as a unit. poly means many. most simple polyatomic ions are anions.
-Molecular: The name and formula of a molecular compound describe the type and number of atoms in a molecule of the compound. The most metallic element appears first in the name. The name of the second element is changed to end in the suffix -ide. for example: Carbon Dioxide.
WRITING CHEMICAL FORMULAS
-Binary and Polyatomic: Place the symbol of the cation first, followed by the symbol of the anion. Use subscripts to show the ratio of the ions in the compound. Because all compounds are neutral, all total charges on the cations and anions must add up to zero.
-Molecular: Write the symbol for the elements in the order that they appear in the name. The prefixes indicate the number of atoms of each element in the molecule. The prefixes appear as subscripts in the formulas. If there is no prefix for an element in the name, there is only one atom of that element in the molecule.
QUESTIONS
1. What is an ionic bond? (see student solutions)
2.What are the two main types of chemical bonds? (see student solutions)
3.What is polyatomic?
4. how do you write a molecular formula?
1. What is an ionic bond? (see student solutions)
2.What are the two main types of chemical bonds? (see student solutions)
3.What is polyatomic?
4. how do you write a molecular formula?
The light bulb application is that chemical bonds are everywhere around us. They help make up a lot of the things we use. Next time you are using some kind of spice or salt, read the label and see if you can figure out which chemical bond is in it.