ACIDS AND BASES
-Acid: a compound that produces hydronium ions when dissolved in water.
-All acids have certain chemical properties that are similar.
-Sour Taste: Foods that taste sour often contain acids. Lemons, apples, oranges for example, all contain citric acid. Dairy products that have spoiled contain butyric acid. Even though many of the foods we eat contain acids, you should never test if something is acid by tasting it.
-Reactivity With Metals: When you use aluminum foil to cover leftovers such as spaghetti, or something that contains tomatoes, the foil usually turns dark. It may also get small holes in it along with the food acquiring a metallic taste. This is because tomatoes contain citric acid, which reacts with metals such as aluminum. That reaction between acid and metal is an example of single displacement.
-Color Changes in Indicators: Indicator: any substance that changes color in the presence of an acid or a base. one of the most common indicators is litmus. Litmus is a dye derived from plants called lichens. Litmus paper is made by coating paper in litmus. Blue litmus paper turns read in the presence of an acid. If you drop an unknown solution onto blue litmus paper and the litmus paper turns red, that solution is an acid.
-Base: A compound that produces hydroxide ions when dissolved in water.
-Like acids, bases have certain chemical and physical properties used to identify them.
-Bitter Taste: For example, unsweetened chocolate. Without the sugar, chocolate is bitter. Many cough syrups and other liquid medicines contain similar bases. Fruit flavorings are commonly used to mask that bitter taste.
-Slippery Feel: Bases feel slippery. Wet soap and many cleaning products are slippery to the touch.
-Color Changes in Indicators: Bases turn red litmus paper blue. The litmus paper will change back to red if you drop an acidic solution on it. Another acid-base indicator is phenolphthalein. It is red in a base and colorless in an acid. Also, flowers such as hydrangeas are indicators. Whatever color they are shows if the soil they are in is acidic or basic.
-Acid: a compound that produces hydronium ions when dissolved in water.
-All acids have certain chemical properties that are similar.
-Sour Taste: Foods that taste sour often contain acids. Lemons, apples, oranges for example, all contain citric acid. Dairy products that have spoiled contain butyric acid. Even though many of the foods we eat contain acids, you should never test if something is acid by tasting it.
-Reactivity With Metals: When you use aluminum foil to cover leftovers such as spaghetti, or something that contains tomatoes, the foil usually turns dark. It may also get small holes in it along with the food acquiring a metallic taste. This is because tomatoes contain citric acid, which reacts with metals such as aluminum. That reaction between acid and metal is an example of single displacement.
-Color Changes in Indicators: Indicator: any substance that changes color in the presence of an acid or a base. one of the most common indicators is litmus. Litmus is a dye derived from plants called lichens. Litmus paper is made by coating paper in litmus. Blue litmus paper turns read in the presence of an acid. If you drop an unknown solution onto blue litmus paper and the litmus paper turns red, that solution is an acid.
-Base: A compound that produces hydroxide ions when dissolved in water.
-Like acids, bases have certain chemical and physical properties used to identify them.
-Bitter Taste: For example, unsweetened chocolate. Without the sugar, chocolate is bitter. Many cough syrups and other liquid medicines contain similar bases. Fruit flavorings are commonly used to mask that bitter taste.
-Slippery Feel: Bases feel slippery. Wet soap and many cleaning products are slippery to the touch.
-Color Changes in Indicators: Bases turn red litmus paper blue. The litmus paper will change back to red if you drop an acidic solution on it. Another acid-base indicator is phenolphthalein. It is red in a base and colorless in an acid. Also, flowers such as hydrangeas are indicators. Whatever color they are shows if the soil they are in is acidic or basic.
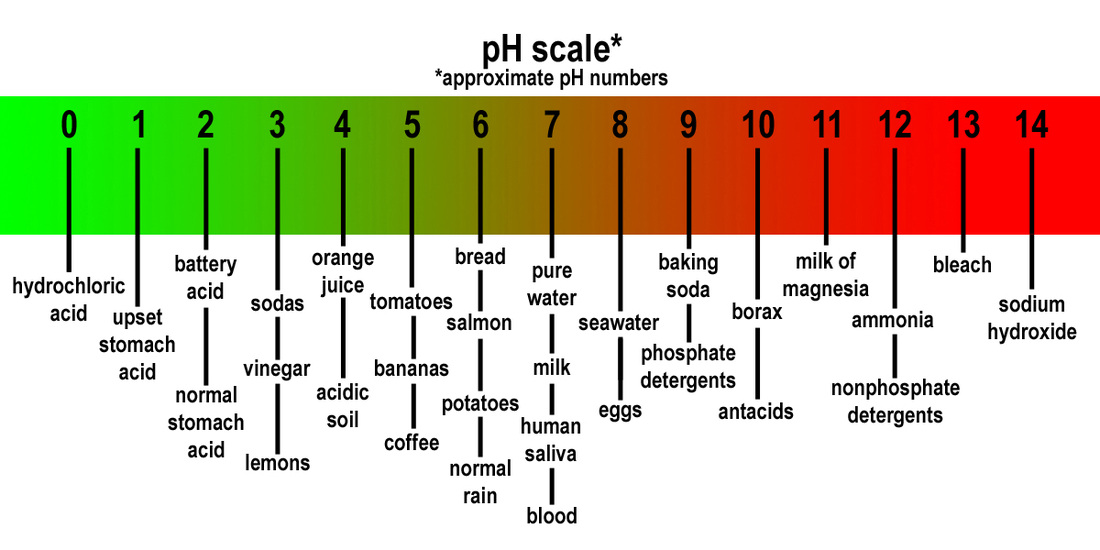
THE PH SCALE
-Chemists use a number scale from 1 to 14 to describe the concentration of hydronium ions in a solution. This is known as the pH scale.
-A pH of 7 indicates a neutral solution. Acids have a pH less than 7. Bases have a pH greater than 7.
-Chemists use a number scale from 1 to 14 to describe the concentration of hydronium ions in a solution. This is known as the pH scale.
-A pH of 7 indicates a neutral solution. Acids have a pH less than 7. Bases have a pH greater than 7.
QUESTIONS
1. What kind of taste do acids have? (see student solutions)
2. Would a pH of 9 be neutral, acidic, or basic? (see student solutions)
3. What does a base feel like?
4. Give two examples of a color change indicator.
1. What kind of taste do acids have? (see student solutions)
2. Would a pH of 9 be neutral, acidic, or basic? (see student solutions)
3. What does a base feel like?
4. Give two examples of a color change indicator.
The light bulb application is the pH scale. Everything has a pH level. Now that you know how to tell if something is basic, acidic, or neutral, the next time you eat something take a guess at what it is!