SCIENTIFIC METHOD
The scientific method is an organized plan for gathering, organizing, and communicating information.
The order of the scientific method is:
1. Observation- using your senses to gather information.
2. Problem- What do you want to know?
3. Hypothesis- Make an educated guess.
4. Experiment- Put you're hypothesis to the test.
5. Analyze Data- Make charts, graphs, reports, anything to show information.
6. Conclusion- Accept or reject you're hypothesis. (If the results are incorrect, form a new hypothesis. TERMS
The scientific method is an organized plan for gathering, organizing, and communicating information.
The order of the scientific method is:
1. Observation- using your senses to gather information.
2. Problem- What do you want to know?
3. Hypothesis- Make an educated guess.
4. Experiment- Put you're hypothesis to the test.
5. Analyze Data- Make charts, graphs, reports, anything to show information.
6. Conclusion- Accept or reject you're hypothesis. (If the results are incorrect, form a new hypothesis. TERMS
- Scientific Theory: This is an explanation based on many observations supported by experimental results.
- Scientific Law: "rule of nature" evidence summed up by related observations and experimental results to describe a pattern found in nature.
- Manipulated Variable (Independent): Causes a change in another variable.
- Responding Variable (Dependent): The variable that changes in response to the manipulated variable.
- Controlled Experiment: A standard for comparison, holds a variable constant.
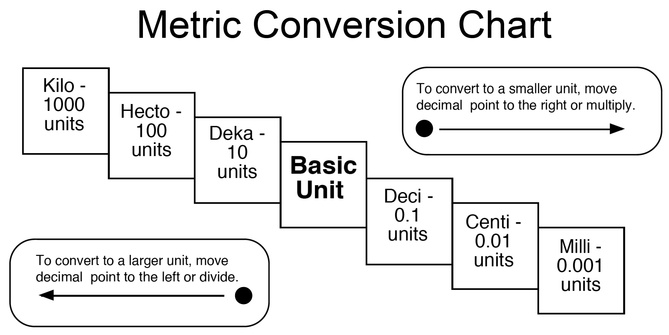
METRIC CONVERSIONS
- A Metric Conversion is the conversion of the same number from one unit to another. for example converting something in centimeters to the same number in millimeters.
-The Units themselves are: Kilo, Hecto, Deka, Base, Deci, Centi, Milli
-You can remember them by this phrase:
"King Henry Died By Drinking Chocolate Milk"
SCIENTIFIC AND STANDARD NOTATION
-Scientific Notation is a way to write a large number in a simpler, smaller way.
-To properly write a number in scientific notation you must multiply:
Whole Number X 10^ exponent
-For example you can take the number 235000000 and write it as ----------- "2.35 X 10^8"
- There are two types of scientific notation. Positive and negative. For example:
2.35 X 10^-8 and 3.97 X 10^-7
-When trying to figure out or "solve" the scientific notation and put it back into standard notation, all you do is start at the decimal and move it certain amount of spaces based on the exponent.
- if the exponent is positive, then you move the decimal to the right. If it's negative, you move it to the left.
-For example: Let's use the scientific notation 2.0 X 10^6. You would start at the decimal after the 2, simply move it over 6 spaces to the right (The exponent is 6), then fill the new spaces in with zeroes. The final answer will come out to 2,000,000.
-If the exponent was a -6 then you would do the exact same process, just to the left instead of the right.
-Standard notation is the form of large numbers we use normally. Such as 2,000,000 instead of
2.0 X 10 ^6.
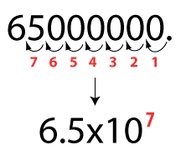
-To convert a number from standard notation to scientific notation, you must first see if the number you are starting with is greater than 1. if it is then the exponent will be positive. If it is not then the exponent will be negative.
- To convert the number properly, you start at the decimal point. Then you count spaces either to the left or to the right, depending on if the number is greater or less than one. You count until you get to one whole number. The decimal will go to the right of that whole number.
-Example:
"2,000,000" Start at the last zero. Then count 6 spaces to the left until you land next to the 2. Put a decimal there along with the number to the right of it, "2.0". Then add "X 10^ positive or negative number of spaces you counted". The final answer will be "2.0 X 10^6"
-Scientific Notation is a way to write a large number in a simpler, smaller way.
-To properly write a number in scientific notation you must multiply:
Whole Number X 10^ exponent
-For example you can take the number 235000000 and write it as ----------- "2.35 X 10^8"
- There are two types of scientific notation. Positive and negative. For example:
2.35 X 10^-8 and 3.97 X 10^-7
-When trying to figure out or "solve" the scientific notation and put it back into standard notation, all you do is start at the decimal and move it certain amount of spaces based on the exponent.
- if the exponent is positive, then you move the decimal to the right. If it's negative, you move it to the left.
-For example: Let's use the scientific notation 2.0 X 10^6. You would start at the decimal after the 2, simply move it over 6 spaces to the right (The exponent is 6), then fill the new spaces in with zeroes. The final answer will come out to 2,000,000.
-If the exponent was a -6 then you would do the exact same process, just to the left instead of the right.
-Standard notation is the form of large numbers we use normally. Such as 2,000,000 instead of
2.0 X 10 ^6.
-To convert a number from standard notation to scientific notation, you must first see if the number you are starting with is greater than 1. if it is then the exponent will be positive. If it is not then the exponent will be negative.
- To convert the number properly, you start at the decimal point. Then you count spaces either to the left or to the right, depending on if the number is greater or less than one. You count until you get to one whole number. The decimal will go to the right of that whole number.
-Example:
"2,000,000" Start at the last zero. Then count 6 spaces to the left until you land next to the 2. Put a decimal there along with the number to the right of it, "2.0". Then add "X 10^ positive or negative number of spaces you counted". The final answer will be "2.0 X 10^6"
MEASUREMENTS
-Many things fall under this category. Scientific and Standard notation are just a couple of them.
Base Units and Derived Units (SI units)
-SI is built on seven metric units, or base units. Below is a list of SI base units.
-Length - meter -m - Time - second - s -Luminous intensity - candela -
-Mass - kilogram -kg - Amount of substance - mole - mol cd--------^
-Temperature - Kelvin - k - Electric current - ampere - A
-There are additional SI units called derived units. They are made from combinations of base units.
-Area - square meter - m^2 -Pressure - pascal - Pa -Electric charge - coulomb - C -Volume - cubic meter - m^3 -Energy - joule - J
-Density - kg per cubic meter - kg/mg^3 -Frequency - hertz - Hz
Vocab
- Length: The straight-line distance between two points.
- Mass: The quantity of matter in an object or sample.
- Volume: The amount of space taken up by an object.
Limits of Measurement
-Precision: a gauge of how exact a measurement is.
-According to an analog clock it might take you 5 minutes to get ready. But a digital clock might say 5 minutes ans 15 seconds. That second measurement has more significant figures.
-Significant Features: All the digits that are known in a measurement, plus the last digit that is estimated.
-The precision of a calculated answer is limited by the least precise measurement used in the calculation.
-Accuracy: The closeness of a measurement to the actual value of what is being measured.
Measuring Temperature
- Thermometer: An instrument that measures temperature, or how hot an object is.
- The degree measure for temperature is Fahrenheit and Celsius.
-A degree Celsius is almost twice as large as a degree Fahrenheit.
-The SI base unit for temperature is Kelvin (K).
- 0k is the lowest possible temperature that can be reached.
-The formula to convert K into Celsius is
K = the degree of Celsius + 273
QUESTIONS
-Scientific Notation is _____________________. (see student solutions)
-How many units is SI built on? (see student solutions)
-Convert 4,000,000 to scientific notation.
-Write the formula used to convert K into Celsius.
-Many things fall under this category. Scientific and Standard notation are just a couple of them.
Base Units and Derived Units (SI units)
-SI is built on seven metric units, or base units. Below is a list of SI base units.
-Length - meter -m - Time - second - s -Luminous intensity - candela -
-Mass - kilogram -kg - Amount of substance - mole - mol cd--------^
-Temperature - Kelvin - k - Electric current - ampere - A
-There are additional SI units called derived units. They are made from combinations of base units.
-Area - square meter - m^2 -Pressure - pascal - Pa -Electric charge - coulomb - C -Volume - cubic meter - m^3 -Energy - joule - J
-Density - kg per cubic meter - kg/mg^3 -Frequency - hertz - Hz
Vocab
- Length: The straight-line distance between two points.
- Mass: The quantity of matter in an object or sample.
- Volume: The amount of space taken up by an object.
Limits of Measurement
-Precision: a gauge of how exact a measurement is.
-According to an analog clock it might take you 5 minutes to get ready. But a digital clock might say 5 minutes ans 15 seconds. That second measurement has more significant figures.
-Significant Features: All the digits that are known in a measurement, plus the last digit that is estimated.
-The precision of a calculated answer is limited by the least precise measurement used in the calculation.
-Accuracy: The closeness of a measurement to the actual value of what is being measured.
Measuring Temperature
- Thermometer: An instrument that measures temperature, or how hot an object is.
- The degree measure for temperature is Fahrenheit and Celsius.
-A degree Celsius is almost twice as large as a degree Fahrenheit.
-The SI base unit for temperature is Kelvin (K).
- 0k is the lowest possible temperature that can be reached.
-The formula to convert K into Celsius is
K = the degree of Celsius + 273
QUESTIONS
-Scientific Notation is _____________________. (see student solutions)
-How many units is SI built on? (see student solutions)
-Convert 4,000,000 to scientific notation.
-Write the formula used to convert K into Celsius.
There are many real life applications to the topics on this page. One of the most profound ones, in my opinion, is metric conversions. You can use that almost anywhere! If you're at the grocery store comparing two items one of which is in grams and the other in milligrams, just use your knowledge of how to convert! You will know just how close in size they are.