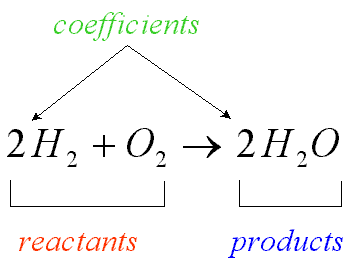CHEMICAL EQUATIONS
-Reactants: The substances that undergo a change.
-Products: New substance formed as a result of a change.
-Using equations to represent reactions: During a chemical reaction, the reactants change into products. this is summarized by a word equation.
Reactants -----> Products
You can substitute the reactants and products of the reaction into the word equation as follows
Carbon + Oxygen ------> Carbon dioxide
then, you can simplify that word equation by writing the reactants and products as chemical formulas.
C + O2 -----> CO2
-Conservation of mass: During chemical reactions, the ass of the products is always equal to the mass of the reactants. This principal was established by a French chemist named Antoine Lavoisier. It is known as the law of conservation of mass. It states that mass is neither created or destroyed in a chemical reaction.
-Reactants: The substances that undergo a change.
-Products: New substance formed as a result of a change.
-Using equations to represent reactions: During a chemical reaction, the reactants change into products. this is summarized by a word equation.
Reactants -----> Products
You can substitute the reactants and products of the reaction into the word equation as follows
Carbon + Oxygen ------> Carbon dioxide
then, you can simplify that word equation by writing the reactants and products as chemical formulas.
C + O2 -----> CO2
-Conservation of mass: During chemical reactions, the ass of the products is always equal to the mass of the reactants. This principal was established by a French chemist named Antoine Lavoisier. It is known as the law of conservation of mass. It states that mass is neither created or destroyed in a chemical reaction.
TYPES OF REACTIONS
-Just as you can classify matter into different types, you can classify chemical reactions into different types. Reactions are often classified by the type of reactant or number of reactants and products.
-Synthesis Reaction: A reaction in which two or more substances react to form a single substance. The reactants can e elements or compounds. The general equation for a synthesis reaction is
A + B ----> AB
-Decomposition Reaction: A reaction in which a compound breaks down into two or more simpler substances. The reactant must be a compound. The products can be elements or compounds. The general equation is
AB -----> A + B
-Single Replacement Reaction: A reaction in which one element takes the place of another element in a compound. These reactions have the general form of
A + BC ------> B + AC
-Double Replacement Reaction: A reaction in which two different compounds exchange positive ions and form two new compounds. The general form is
AB + CD -------> AD + CB
-Combustion Reaction: A reaction in which a substance reacts rapidly with oxygen, often producing heat and light. The burning of a natural gas is an example. The main component of natural gas is methane, CH4. When methane burns in an unlimited supply of oxygen, the following reaction occurs.
CH4 + 2O2 -----> CO2 + 2H2O
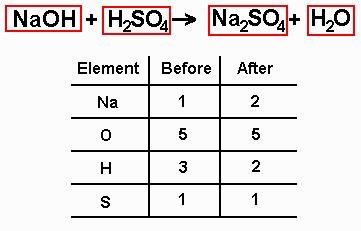
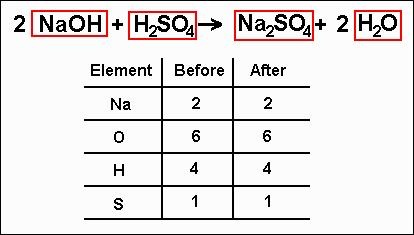
BALANCING EQUATIONS
-In order to show that mass is conserved during a reaction, a chemical equation must be balanced.
-You can balance a chemical equation by changing the coefficients, or the numbers that are before the formulas.
-When you change a coefficient, you change the amount of that reactant or product represented in the chemical equation.
-Never change the subscripts as you balance the equation.
-Changing the formula changes the identity of that reactant or product.
-Step 1- Count the number of atoms of each element on each side of the equation.
-step 2- Change one or more coefficients until the equation is balanced.
-When I was in third grade I hid from my teacher in the slide during recess everyday because she scared me.
-After those two steps, the equation is balanced.
-Just as you can classify matter into different types, you can classify chemical reactions into different types. Reactions are often classified by the type of reactant or number of reactants and products.
-Synthesis Reaction: A reaction in which two or more substances react to form a single substance. The reactants can e elements or compounds. The general equation for a synthesis reaction is
A + B ----> AB
-Decomposition Reaction: A reaction in which a compound breaks down into two or more simpler substances. The reactant must be a compound. The products can be elements or compounds. The general equation is
AB -----> A + B
-Single Replacement Reaction: A reaction in which one element takes the place of another element in a compound. These reactions have the general form of
A + BC ------> B + AC
-Double Replacement Reaction: A reaction in which two different compounds exchange positive ions and form two new compounds. The general form is
AB + CD -------> AD + CB
-Combustion Reaction: A reaction in which a substance reacts rapidly with oxygen, often producing heat and light. The burning of a natural gas is an example. The main component of natural gas is methane, CH4. When methane burns in an unlimited supply of oxygen, the following reaction occurs.
CH4 + 2O2 -----> CO2 + 2H2O
BALANCING EQUATIONS
-In order to show that mass is conserved during a reaction, a chemical equation must be balanced.
-You can balance a chemical equation by changing the coefficients, or the numbers that are before the formulas.
-When you change a coefficient, you change the amount of that reactant or product represented in the chemical equation.
-Never change the subscripts as you balance the equation.
-Changing the formula changes the identity of that reactant or product.
-Step 1- Count the number of atoms of each element on each side of the equation.
-step 2- Change one or more coefficients until the equation is balanced.
-When I was in third grade I hid from my teacher in the slide during recess everyday because she scared me.
-After those two steps, the equation is balanced.
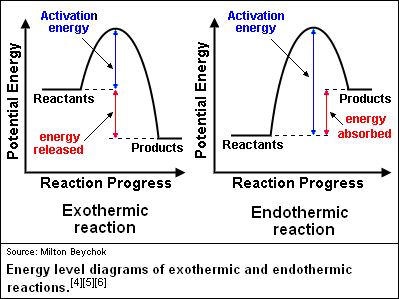
ENDOTHERMIC AND EXOTHERMIC REACTIONS
-Exothermic Reaction: A chemical reaction that releases energy into its surroundings.
-In these reactions the energy the energy released as the products form is greater than the energy required to break the bonds in the reactants.
-In any reaction, the chemical energy reaches a peak before the reactants change into products. That peak represents the amount of energy needed to break the chemical bond of the reactants.
-Endothermic Reaction: A chemical reaction that absorbs energy from its surroundings.
-More energy is required to break the bonds in the reactants than is released by the formation of the products.
-Exothermic Reaction: A chemical reaction that releases energy into its surroundings.
-In these reactions the energy the energy released as the products form is greater than the energy required to break the bonds in the reactants.
-In any reaction, the chemical energy reaches a peak before the reactants change into products. That peak represents the amount of energy needed to break the chemical bond of the reactants.
-Endothermic Reaction: A chemical reaction that absorbs energy from its surroundings.
-More energy is required to break the bonds in the reactants than is released by the formation of the products.
QUESTIONS
1. What is a reactant? (see student solutions)
2. What type of reaction is 2Na + Cl2 -----> 2NaCL? (see student solutions)
3.Write an example of a single replacement reaction.
4. Balance the equation Cu + O2 -----> CuO.
1. What is a reactant? (see student solutions)
2. What type of reaction is 2Na + Cl2 -----> 2NaCL? (see student solutions)
3.Write an example of a single replacement reaction.
4. Balance the equation Cu + O2 -----> CuO.
The light bulb application is endothermic and exothermic reactions. Our bodies are perfect examples of that. We take in energy from food and we give out energy through work. See if you can find examples of how our bodies are endothermic and exothermic.