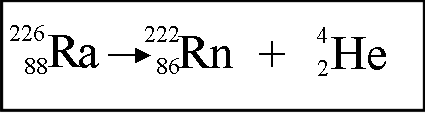RADIOACTIVITY
-Radioactivity: The process in which an unstable atomic nucleus emits charged particles and energy.
-An atom containing an unstable nucleus is called a radioactive isotope, or radioisotope for short.
-During nuclear decay, atoms of one element can change into atoms of a different element.
-Nuclear Radiation: Charged particles and energy that are emitted from the nuclei of radioisotopes.
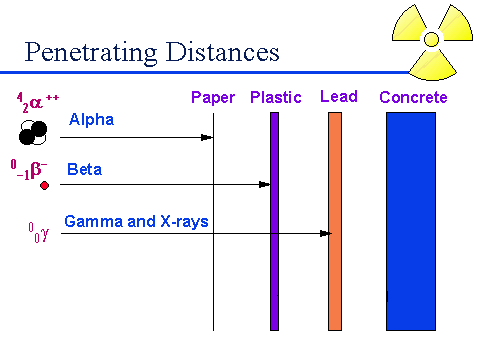
-Alpha Decay: An alpha particle is a positively charged particle made up of two protons and two neutrons. his is an example of a nuclear reaction. Alpha particles are the least penetrating type of nuclear radiation. They travel no more than a few centimeters and can be stopped by paper or clothes.
-Radioactivity: The process in which an unstable atomic nucleus emits charged particles and energy.
-An atom containing an unstable nucleus is called a radioactive isotope, or radioisotope for short.
-During nuclear decay, atoms of one element can change into atoms of a different element.
-Nuclear Radiation: Charged particles and energy that are emitted from the nuclei of radioisotopes.
-Alpha Decay: An alpha particle is a positively charged particle made up of two protons and two neutrons. his is an example of a nuclear reaction. Alpha particles are the least penetrating type of nuclear radiation. They travel no more than a few centimeters and can be stopped by paper or clothes.
-Beta Decay: A beta particle is an electron emitted by an unstable nucleus. Because of its negative charge, a beta particle has an atomic number of -1. It also has a mass number or 0. Beta particles are more penetrating than alpha particles. They can pass through paper, but are stopped by a thin sheet of metal.
-Gamma Decay: A gamma ray is a penetrating ray of energy emitted by an unstable nucleus. They have no mass and no charge. They can travel through space at the speed of light. They are extremely penetrating. It takes several centimeters of lead or or several meters of concrete to stop gamma radiation.
-Gamma Decay: A gamma ray is a penetrating ray of energy emitted by an unstable nucleus. They have no mass and no charge. They can travel through space at the speed of light. They are extremely penetrating. It takes several centimeters of lead or or several meters of concrete to stop gamma radiation.
FISSION
-Fission: The splitting of an atomic nucleus into two smaller parts.
-In nuclear fission, tremendous amounts of energy can be produced from very small amounts of mass.
-Fission causes chain reactions. Whenever a fission happens, it reacts with surrounding things and there is just fission everywhere.
-In a chain reaction, neutrons released during the splitting of an initial nucleus trigger series of chain fissions.
-Critical Mass: The smallest possible mass of a fissionable material that can sustain a chain reaction.
-Nowadays about 20 percent of electricity in America comes from nuclear power plants.
-Fission: The splitting of an atomic nucleus into two smaller parts.
-In nuclear fission, tremendous amounts of energy can be produced from very small amounts of mass.
-Fission causes chain reactions. Whenever a fission happens, it reacts with surrounding things and there is just fission everywhere.
-In a chain reaction, neutrons released during the splitting of an initial nucleus trigger series of chain fissions.
-Critical Mass: The smallest possible mass of a fissionable material that can sustain a chain reaction.
-Nowadays about 20 percent of electricity in America comes from nuclear power plants.
FUSION
-Fusion: a process in which the nuclei of two atoms combine to form a larger nucleus.
-Fusion requires extremely high temperatures.
-Usually where fusion can naturally happen, matter exists as plasma.
-Scientists who want to make a fusion reactor come across two problems.
- Getting those hot temperatures, and containing the plasma.
HALF-LIFE
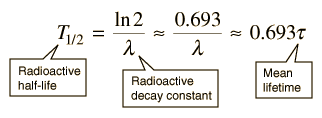
-Half Life: The time required for one half of a sample of a radioisotope to decay.
-Half lives can vary from fractions of a second, to billions of years.
-Unlike chemical reaction rates, which vary with condition of reaction, nuclear decay rates are constant.
-Fusion: a process in which the nuclei of two atoms combine to form a larger nucleus.
-Fusion requires extremely high temperatures.
-Usually where fusion can naturally happen, matter exists as plasma.
-Scientists who want to make a fusion reactor come across two problems.
- Getting those hot temperatures, and containing the plasma.
HALF-LIFE
-Half Life: The time required for one half of a sample of a radioisotope to decay.
-Half lives can vary from fractions of a second, to billions of years.
-Unlike chemical reaction rates, which vary with condition of reaction, nuclear decay rates are constant.
QUESTIONS
1. What is radioactivity? (see student solutions)
2. What is the half life of lead? (see student solutions)
3. What happens when fission takes place?
4. What percent of electricity in America comes from nuclear power plants?
1. What is radioactivity? (see student solutions)
2. What is the half life of lead? (see student solutions)
3. What happens when fission takes place?
4. What percent of electricity in America comes from nuclear power plants?
The light bulb application is nuclear energy. Nuclear energy is a very cool thing. We make bombs with it, we make electricity with it, and many other things. There is so much to learn about all things nuclear.