ATOMIC STRUCTURE
ATOMS
-Atom: The smallest particle of an element.
-There have been many different models of the atom throughout history. An example of one is from the Ancient Greeks.
-Ancient Greek Model- Philosopher Democritus believed everything was made up of small particles that could not be divided. He named those particles atoms which comes from the Greek word atomos which means "uncut".
-There have also many atomic theories from various individuals.
-Dalton's Atomic Theory: John Dalton was a English teacher who lived in the 17-1800s. He spent a lot of his spare time doing different science experiments. He proposed the theory that all matter is made up of individual particles called atoms, which cannot be divided. The main points in that theory are:
-All elements are composed of atoms.
-All atoms of the same element have the same mass, and all atoms of different elements have different masses.
-Compounds contain atoms of more than one element.
-In a particular compound, atoms of different elements always combine in the same way.
-For his model, he made wooden spheres to represent the atoms of different elements.
-Another is Rutherford's theory.
-Ernest Rutherford lived in the 18-1900s. Through various experiments he discovered the nucleus.
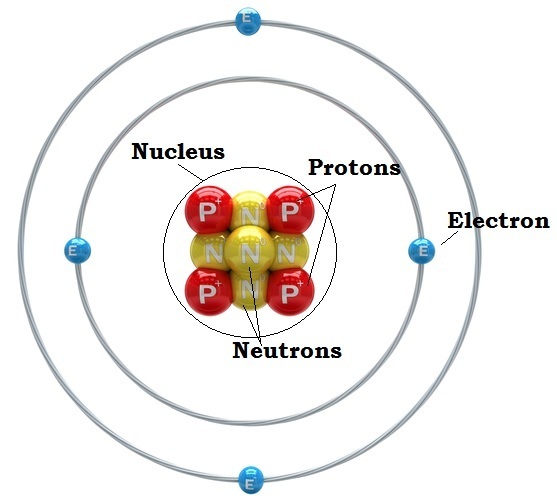
-Nucleus: A dense positively charged mass located in the center of the atom.
-According to Rutherford's model, all of an atoms positive charge is concentrated in its nucleus.
ATOMS
-Atom: The smallest particle of an element.
-There have been many different models of the atom throughout history. An example of one is from the Ancient Greeks.
-Ancient Greek Model- Philosopher Democritus believed everything was made up of small particles that could not be divided. He named those particles atoms which comes from the Greek word atomos which means "uncut".
-There have also many atomic theories from various individuals.
-Dalton's Atomic Theory: John Dalton was a English teacher who lived in the 17-1800s. He spent a lot of his spare time doing different science experiments. He proposed the theory that all matter is made up of individual particles called atoms, which cannot be divided. The main points in that theory are:
-All elements are composed of atoms.
-All atoms of the same element have the same mass, and all atoms of different elements have different masses.
-Compounds contain atoms of more than one element.
-In a particular compound, atoms of different elements always combine in the same way.
-For his model, he made wooden spheres to represent the atoms of different elements.
-Another is Rutherford's theory.
-Ernest Rutherford lived in the 18-1900s. Through various experiments he discovered the nucleus.
-Nucleus: A dense positively charged mass located in the center of the atom.
-According to Rutherford's model, all of an atoms positive charge is concentrated in its nucleus.
CALCULATING PROTONS ELECTRONS AND NEUTRONS
-Every atom has three key things inside of it. Protons, Electrons, and Neutrons.
-Electrons have a negative charge and are located around the nucleus.
-To find the number of electrons look at the atoms atomic number.
-Protons have a positive charge and are located inside the nucleus.
-To find the number of protons look at the atoms atomic number.
-Neutrons have no charge and are located inside of the nucleus.
-To find the number of neutrons, Mass# - Atomic#
ELECTRON CONFIGURATION
-Configuration: An arrangement of objects in a given space.
-Electron Configuration: The arrangement of electrons in the orbitals of an atom.
-The most stable electron configuration is the one in which the electrons are in orbitals with the lowest possible energies.
BOHR MODEL
-Bohr believed that the nucleus contained the protons and neutrons but the electrons orbited in different energy levels, ore shells, around it.
-Every atom has three key things inside of it. Protons, Electrons, and Neutrons.
-Electrons have a negative charge and are located around the nucleus.
-To find the number of electrons look at the atoms atomic number.
-Protons have a positive charge and are located inside the nucleus.
-To find the number of protons look at the atoms atomic number.
-Neutrons have no charge and are located inside of the nucleus.
-To find the number of neutrons, Mass# - Atomic#
ELECTRON CONFIGURATION
-Configuration: An arrangement of objects in a given space.
-Electron Configuration: The arrangement of electrons in the orbitals of an atom.
-The most stable electron configuration is the one in which the electrons are in orbitals with the lowest possible energies.
BOHR MODEL
-Bohr believed that the nucleus contained the protons and neutrons but the electrons orbited in different energy levels, ore shells, around it.
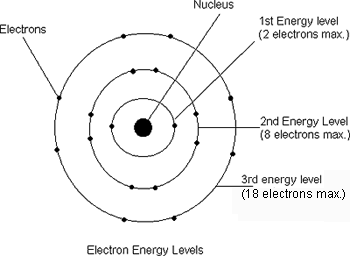
-The Bohr model is a chart that allows us to show the number of electrons in the shells of the nucleus.
-The first shell holds 2 electrons.
-The second shell holds 8 electrons.
-The third shell holds 18 electrons.
-Each shell holds more and more electrons as you go out, although you rarely go above three shells.
-To make a Bohr model of an element, say that has 5 electrons, you would start at the closest shell to the nucleus and start filling them in. Two electron dots would go in the first shell. After that there is still three electrons left that need to be shown. because the first shell can only hold two electrons, and is full, we move to the next shell. The second shell holds eight electrons. So you can simply fill in the rest of your electron dots in that second shell.
-If you were doing a problem with a large number of electrons, just keep filling in the shells. When one gets full, move to the next one until you have shown all the electrons in that element!
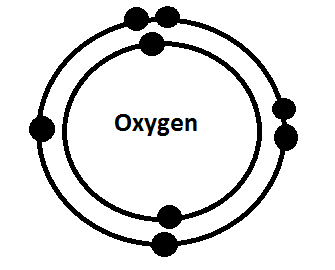
-The picture below is an example of a Bohr model for Oxygen.
-The first shell holds 2 electrons.
-The second shell holds 8 electrons.
-The third shell holds 18 electrons.
-Each shell holds more and more electrons as you go out, although you rarely go above three shells.
-To make a Bohr model of an element, say that has 5 electrons, you would start at the closest shell to the nucleus and start filling them in. Two electron dots would go in the first shell. After that there is still three electrons left that need to be shown. because the first shell can only hold two electrons, and is full, we move to the next shell. The second shell holds eight electrons. So you can simply fill in the rest of your electron dots in that second shell.
-If you were doing a problem with a large number of electrons, just keep filling in the shells. When one gets full, move to the next one until you have shown all the electrons in that element!
-The picture below is an example of a Bohr model for Oxygen.
QUESTIONS
1.What is an atom? (see student solutions)
2.How many neutrons are in an element if its mass number is 20 and its atomic number is 10? (see student solutions)
3.How many electrons does the second shell hold?
4.Draw a Bohr model for gold.
1.What is an atom? (see student solutions)
2.How many neutrons are in an element if its mass number is 20 and its atomic number is 10? (see student solutions)
3.How many electrons does the second shell hold?
4.Draw a Bohr model for gold.
The light bulb application here is atoms. Every single thing has atoms. If you just think about how many atoms could possibly be in your body or in whatever is around you, it's mind boggling! Without atoms, those tiny little things that we cant even see, we could not live.